Self-Assembling Acrylic Block Copolymers for Enhanced Adhesives Properties
A new type of acrylic copolymer has been developed that features block structures and controlled functional group placement due to a controlled radical polymerization process.
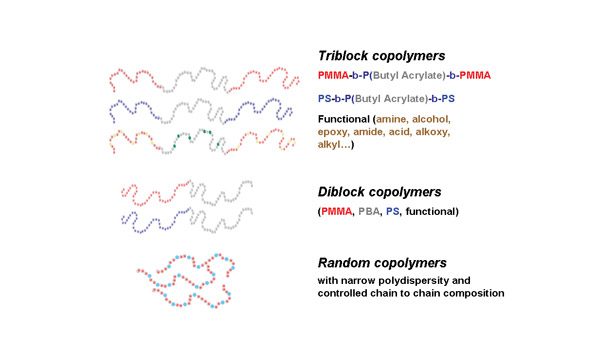

Figure 2. Atomic Force Microscopy of Two Types of Morphologies Obtained with a Block Copolymer Having 60% of Soft Block (left) and 85% of Soft Block (right)

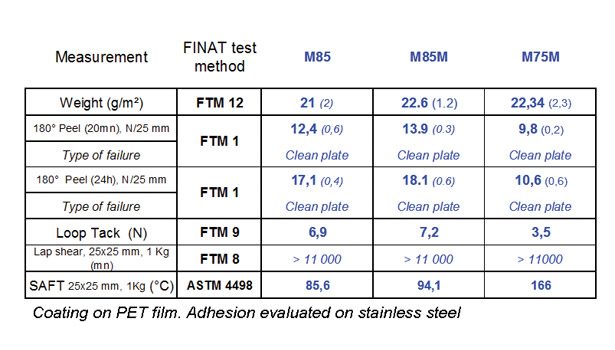
Table 1. Physical properties of pure Nanostrength® block copolymers.
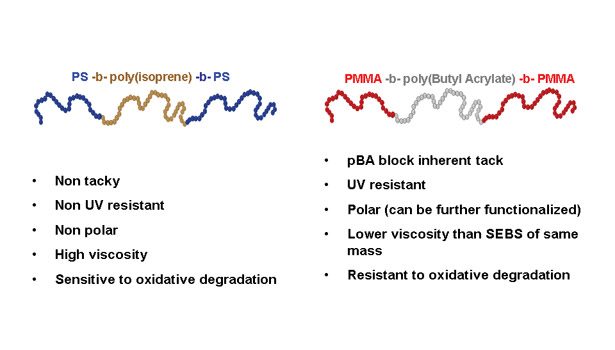
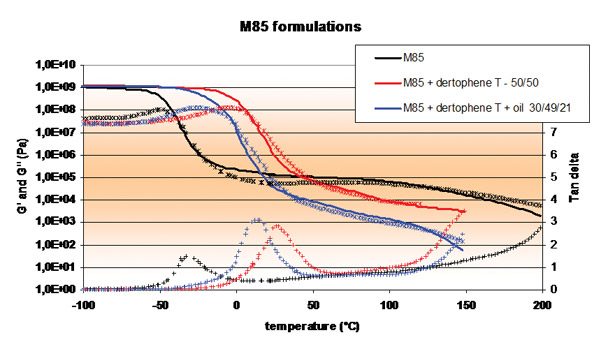
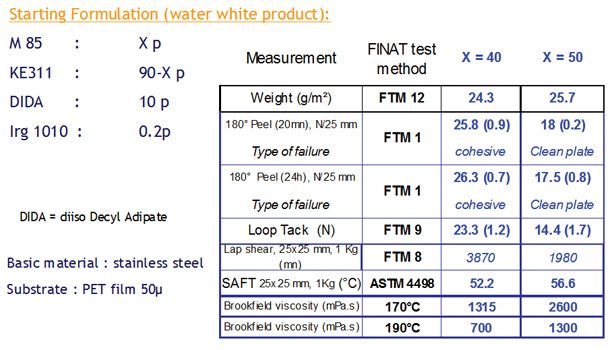
Figure 4. Nanostrength M85-Based Formulations with Different Additives







Acrylic polymers are widely used in pressure-sensitive adhesive (PSA) applications in the form of emulsion, solvent or radiation-cured systems. They combine good UV resistance, transparency, tailored tack, and stiffness due to a large choice of acrylic and methacrylic comonomers.
Acrylic copolymers are produced through conventional free radical polymerization processes that lead to random structures with broad polydispersities. In addition, free radical polymerization does not allow for the introduction of functional groups at specific locations along the polymer chains. For adhesive applications, a key synthesis challenge is the optimization of the polymer composition and molecular weight to achieve low viscosities, good adhesion, and creep resistance.
This balance of antagonistic tack/creep properties is generally achieved through extremely high molecular weights, often above 1 million g/mol, or partial crosslinking. However, both options are detrimental for lamination properties and flow behavior. Extremely high molecular weights prevent their use in hot-melt adhesives, while crosslinking can require several days to be completed. UV-curable acrylic adhesives are another alternative for high-performance adhesives, but their implementation requires significant equipment investment and expertise.
A new type of acrylic copolymer* has been developed that features block structures and controlled functional group placement due to a controlled radical polymerization (CRP) process. These polymers bring a good balance of tack and creep properties in the absence of crosslinking, which enables their use in acrylic hot-melt or solventborne adhesives.
*Introduced by Arkema under the trademark Nanostrength®.
Controlled Radical Polymerization
The new block copolymers for PSAs consist in a hard-soft-hard triblock structure, alternating a high Tg methyl methacrylate block with a low Tg butyl acrylate block. The synthesis of these new structures was enabled by controlled radical polymerization (CRP) and, more precisely, nitroxide-mediated polymerization (NMP) based on a proprietary alkoxyamine.** This commercially available molecule combines a polymerization initiator and a radical trap. The radical stabilization provided by the nitroxide allows the controlled polymerization of a “living” first block (A) that is then used as a macro-initiator for the polymerization of the two external blocks (B), leading to a BAB structure.
NMP’s main advantage over other CRP technologies, such as ATRP, is that the polymers are free of any metal impurities. In addition, the alkoxyamine is a versatile initiator/controller that can be used in bulk, emulsion, solvent and suspension processes. It controls a broad range of monomer types, including styrenics, almost all types of acrylates and (under specific conditions) methacrylates. The extremely large range of available functional acrylic and methacrylic monomers offers the possibility to selectively introduce specific functional groups into the blocks. Typical structures achievable with this technology include triblock, diblock or random block copolymers, as illustrated in Figure 1.
As mentioned previously, the newly developed block copolymers consist in a P(methyl methacrylate)-b-P(butyl acrylate)-b-P(methyl methacrylate) sequence. Due to the immiscibility between the two types of blocks, the block copolymers self-assemble into soft domains, providing tack and low temperature elasticity. Hard domains behave as physical crosslinks, bringing cohesion and mechanical properties. The morphology of the various phases depends on the ratio between the hard and soft blocks, the length of the blocks, and temperature. Figure 2 illustrates two types of morphologies, one lamellar and one micellar, that can be achieved by varying the amount of soft block.
**Arkema’s BlocBuilder® alkoxyamine.
Block Copolymer Structures
The new block copolymers for PSAs contain 70-85% of soft poly(butyl acrylate). In addition, they have been synthesized in two versions, a non-functional version and one that has been modified with acid groups in the hard PMMA end-blocks.† The presence of acid raises the Tg of the end-block and improves performance at elevated temperature, as illustrated in Table 1 by the higher G’-G’’ crossover temperature of M85M compared to M85. The presence of acid groups also enhances phase separation between soft PBA and hard PMMA domains. Finally, these acid groups can also be used as reactive sites for further chemical reactions (e.g., controlled crosslinking through the addition of molecules containing epoxy groups, amines, etc.). Product characteristics are given in Table 1.
Due to their nanostructuration, these polymers have excellent transparencies even when used as a non-formulated material, and have good elastomeric properties with high stress at break values even for the softest grades. Styrene-acrylate-styrene triblock copolymer (SAS) can also be obtained.
†Nanostrength® M75 and M85, and Nanostrength® M75M and M85M, respectively.
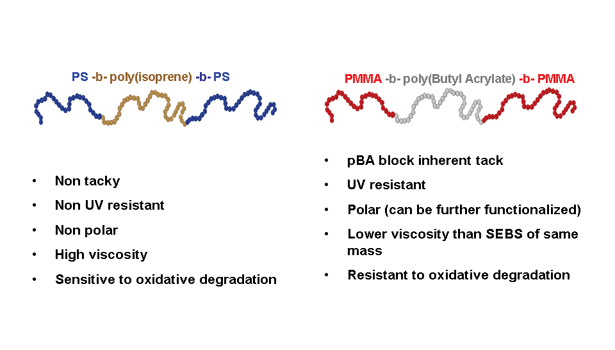
A New Class of Elastomeric Adhesives?
The so-called “electrometric adhesives” or “elastomeric hot melts” often refer to formulations based on styrenic block copolymers like styrene-isoprene-styrene (SIS) or styrene-ethylene-butylene-styrene (SEBS), and their combinations with their diblock forms. The nanostructuration of these materials is well-known, based again on hard and soft blocks immiscibility.
Since the neat polymers are not tacky, SIS and SEBS adhesives are formulated with combinations of tackifiers and plasticizers, as well as other additives. As opposed to isoprene or ethylenebutylene mid-blocks, soft acrylates like butyl acrylate, 2-ethylhexylacrylate, and octylacrylate, have a higher entanglement molecular weight, leading to lower modulus and inherent tackiness.
The new block copolymers with high levels of PBA can thus be used as elastomeric adhesives with little or no formulation. This can be of particular benefit in applications such as removable adhesives, where the adhesive must be removed without leaving any residue on the substrate surface. Major points of differentiation between SIS and MAM adhesives are summarized in Figure 3.
Adhesion Properties and Processing
Adhesion and creep behavior of the new block copolymers are directly related to the ratio between the soft and hard blocks. The softest products (e.g., M85 and M85M) offer a fair adhesion to a variety of supports. Shear resistance is much higher than what can be achieved with acrylic random copolymers of similar composition and similar molecular weight. Table 2 shows adhesion properties of the pure block copolymers.
Due to their relatively high viscosities in the molten stage, preferred application systems are solution-based or extrusion. Many solvents can be used to dissolve the new block copolymers, including ketones, toluene, xylene, acetates and chlorinated solvents. When the ratio between the soft and hard phase decreases, tack gets very low, but mechanical and high-temperature properties significantly increase, making the block copolymers suitable for structural adhesive applications.
PSA Formulations
Many tackifiers are non-polar molecules and have only limited compatibility with polar acrylates. In general, an ideal tackifier should be selective of the mid-block and not affect the hard structuring block. Various tackifiers and plasticizers for soft block compatibility have been screened. Trends are given below (verification should be performed for each commercial additive and each grade of block copolymer):
• Compatible tackifiers—(hydrogenated) rosin esters, (hydrogenated) aromatic hydrocarbon reins, aromatic modified hydrocarbon resins, aromatic modified polyterpene resins
• Non compatible tackifiers—terpene-styrene, polyterpene resins, aliphatic and cycloaliphatic resins
• Compatible plasticizers—phthalates, adipates, citrates, trioctyltrimellitate, polyethers, fatty acid esters
• Non-compatible plasticizers—mineral oils, paraffin
Figure 4 and Table 3 illustrate potential adhesive properties achievable through formulation. In this case, M85 has been formulated with varied amounts of tackifier and/or plasticizer. It is clear that a broad spectrum of adhesion/creep values could be covered. Higher creep could still be achieved with higher levels of polymers, as viscosities remain reasonably low in the case of M85.
Conclusions
Thanks to controlled radical polymerization, new acrylic block copolymers with interesting adhesive properties have been obtained. Their block structure and their key properties make their use in hot-melt adhesive applications very interesting. Key properties include transparency, UV resistance and intrinsic tack. They are also easy to formulate and can be functionalized to incorporate reactive sites. They possess fully thermoplastic behavior, which makes them removable without residues and easy to color while offering good adhesion onto polar substrates.
The new block copolymers can also be used in solvent-based systems, without the need for crosslinking to achieve good creep resistance. The softest grades already provide an efficient tackiness/creep resistance combination, allowing their use without any formulation, thus avoiding the risks of small molecules migration or residues when used as removable or repositionable adhesives.
For more information, contact the lead author at (610) 878 6948 or mickael.havel@arkema.com, or visit www.arkema.com.
Author’s note: Technical work highlighted in this presentation was mostly generated at Arkema’s R&D center in Lacq, France. Nanostrength® and BlocBuilder® are registered trademarks of Arkema.
To find suppliers of acrylic block copolymers and other products, visit www.adhesivesmag.com/suppliers.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!